Quality Control
Quality Control
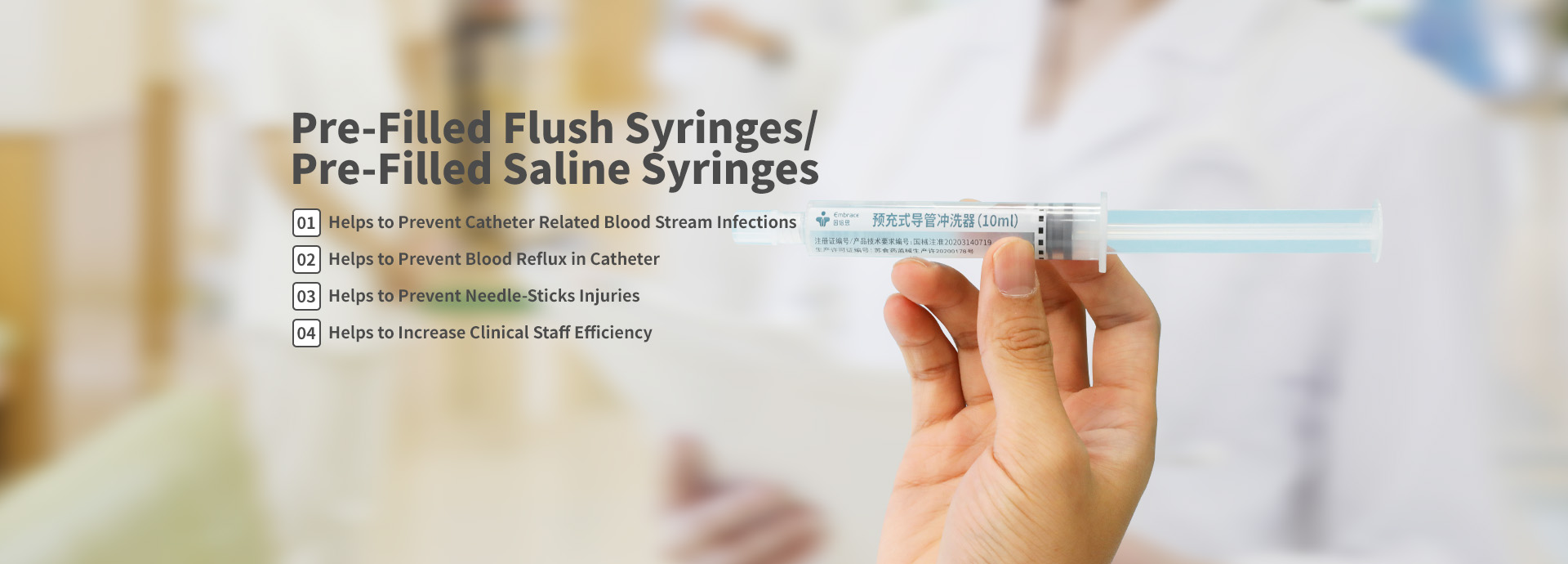
Embrace effective Quality Assurance system ensures that there are robust controls in place that govern the manufacture, testing, storage and distribution of Embrace pre-filled syringes, ensuring strict compliance with its specifications over the product’s shelf life to maximise patient outcomes.
The company's quality policy is highly quality-oriented, safe, effective and customer centric with aim of continuous improvement.
In-order to meet the quality policies and objectives, the company has established a sound organization. The company has established the main control procedures in strict accordance with the pharmaceutical GMP, medical device GMP and ISO 13485 management regulations, including: product, quality system change notification control procedures, medical device main document control procedures, document control procedures, record control procedures, production control procedures, human resources Resource control procedures, design and development control procedures, procurement control procedures, sterile medical device sterilization process validation control procedures, traceability control procedures, product release control procedures, nonconforming product control procedures, etc.
Our company having State-of-the-art automated manufacturing processes significantly minimise potential for contamination, company consists of a high profile central laboratory, sample management room, chemical analysis room, physical and chemical room, standard liquid calibration room, balance room, common instrument room, gas phase chamber, infrared chamber, liquid chromatography room, micro particle endotoxin detection room, high greenhouse, microbial limit detection Room, sterile examination room, positive control room, preparation room, stability test room, sample observation room, etc.